How to Perform Cytotoxicity Testing for PET Heat Shrink Tubing Using ISO 10993-5
You need a good way to do cytotoxicity testing for PET heat shrink ISO 10993-5. Using set steps helps you get correct results. Cleanroom conditions keep your samples safe from germs. L-929 mouse fibroblast cells help you see how cells react during extraction, exposure, and checking. Rules help you test medical devices the right way.
Regulatory Standard | Description |
|---|---|
USP Class VI | Needs tests to check if device materials are safe. |
ISO 10993 | Lists tests based on how long and where the body touches the device. |
FDA Title 21 | Makes sure materials are safe and not harmful to people. |
Good controls and records make your results true and help you follow the rules.
Key Takeaways
Follow ISO 10993-5 rules to make sure testing is safe for PET heat shrink tubing.
Work in a cleanroom to stop germs from getting into the cytotoxicity test.
Keep good records of what you do and what happens to prove your testing is correct.
Use L-929 mouse fibroblast cells to check if the cells are harmed.
Make sure at least 70% of the cells live in the test to show the PET tubing is not toxic to cells.
Preparation for Cytotoxicity Testing
Materials and Equipment
You need the right things to start cytotoxicity testing for PET heat shrink ISO 10993-5. Get these items before you begin:
PET heat shrink tubing samples
Extraction vehicles like cell culture medium or saline
L-929 mouse fibroblast cells
Sterile glassware and pipettes
Cell culture plates
Incubator set to 37°C
Personal protective equipment (PPE)
Positive and negative control materials
A checklist helps you keep track of what you need. You can use a table to check off your supplies:
Item | Ready to Use? |
|---|---|
PET Heat Shrink Tubing | ☐ |
Extraction Vehicle | ☐ |
L-929 Cells | ☐ |
Control Materials | ☐ |
Sterile Equipment | ☐ |
PET Heat Shrink Tubing Sample Prep
Cut the PET heat shrink tubing into small pieces. Wash each piece with sterile water to get rid of dust or oil. Dry the samples in a clean place. Weigh the tubing so you know how much you will use. Put the pieces in a clean container. Write the sample name and date on each container.
Tip: Always wear gloves and use clean tools when touching samples. This helps keep your test results correct.
Controls and Cleanroom Setup
Work in a cleanroom or under a laminar flow hood. This keeps germs and dirt away from your samples. Put all your things on a clean table. Use positive controls, which are known toxic materials, and negative controls, which are known safe materials. Controls show if your test is working right.
Keep a logbook to write down every step you do. Write the date, time, and who did each job. Good notes help prove you followed the right steps for cytotoxicity testing for PET heat shrink ISO 10993-5.
Extraction Process per ISO 10993-5

Extraction Conditions and Media
You need to follow strict rules when you prepare extracts for cytotoxicity testing for PET heat shrink ISO 10993-5. The extraction process helps you find out if any harmful chemicals come out of the tubing. You must use the right extraction media. Most labs use cell culture medium, like MEM or DMEM, because these support cell growth. Sometimes, you may use saline or other fluids, depending on your test plan.
Set the extraction ratio by using 6 cm² of PET heat shrink tubing for every 1 mL of extraction medium. If you measure by weight, use 0.2 grams of tubing per 1 mL of medium. Keep the samples in a glass or plastic container that does not react with your test materials.
Note: Always use the same type of extraction media and ratio as other labs. This helps you compare your results with theirs.
Performing the Extraction
Place your prepared PET heat shrink tubing samples into the container with the extraction medium. Seal the container tightly. Put the container in an incubator set to 37°C. Let the samples sit for 24 hours. This temperature and time match the conditions inside the human body.
After 24 hours, remove the container from the incubator. Carefully collect the extract. Do not let any tubing pieces get into the liquid. Use a sterile filter if you need to remove small particles. Label the extract with the sample name, date, and extraction details.
You must follow these steps each time you do cytotoxicity testing for PET heat shrink ISO 10993-5. This makes sure your results are reliable and can be checked by others. Good extraction methods help you meet ISO 10993-5 rules and keep your tests fair.
Cytotoxicity Testing for PET Heat Shrink ISO 10993-5
Cell Culture Setup
You need healthy cells to get good results in cytotoxicity testing for PET heat shrink ISO 10993-5. L-929 mouse fibroblast cells work best for this test. You should grow these cells in a special medium. The table below shows what you need for the cell culture:
Component | Concentration/Condition |
|---|---|
Culture Medium | Minimum Essential Medium (MEM) |
Horse Serum (HS) | 10% |
Penicillin-Streptomycin (P/S) | 1% |
HEPES solution | 1% |
MEM Non-essential Amino Acid Solution | 1% (100×) |
Sodium Pyruvate solution | 1% |
GlutaMAXTM-1 | 1% (100×) |
Incubation Temperature | 37 °C |
CO2 Concentration | 5% |
Medium Replacement Frequency | Every three days |
You should seed the cells at the right density. For most tests, you can use 5,000 cells per square centimeter or 50,000 cells per well. Place the cells in a 96-well plate or another suitable plate. Make sure the cells cover the bottom of each well. Keep the cells in an incubator at 37°C with 5% CO2. Change the medium every three days to keep the cells healthy.
Tip: Check the cells under a microscope. Healthy cells look spread out and stick to the plate.
Exposure of Cells to Extracts
You need to expose the cells to the extracts from your PET heat shrink tubing. First, remove the old medium from each well. Add the extract to the wells with the cells. Use enough extract to cover the cells. You should also set up control wells. Add a positive control (a known toxic substance) to some wells. Add a negative control (a known safe substance) to other wells. This helps you see if your test works as it should.
You can use the following setup for your plate:
Test wells: Add PET heat shrink extract
Positive control wells: Add toxic control extract
Negative control wells: Add safe control extract
Note: Always label your plate and record which wells have which samples.
Incubation and Controls
After you add the extracts, put the plate back in the incubator. Keep the cells at 37°C with 5% CO2. Let the cells incubate for 24 to 48 hours. This time lets you see if the extract harms the cells. Do not disturb the plate during this time.
You must include both positive and negative controls in every test. The positive control should cause cell death. The negative control should not harm the cells. If your controls work, you can trust your test results.
Check the cells after incubation. Look for changes in cell shape, color, or number. Write down your observations. Good records help you show that you followed the right steps for cytotoxicity testing for PET heat shrink ISO 10993-5.
Reminder: Always follow your lab’s safety rules when handling cells and extracts. Wear gloves and keep your work area clean.
Evaluation and Data Analysis
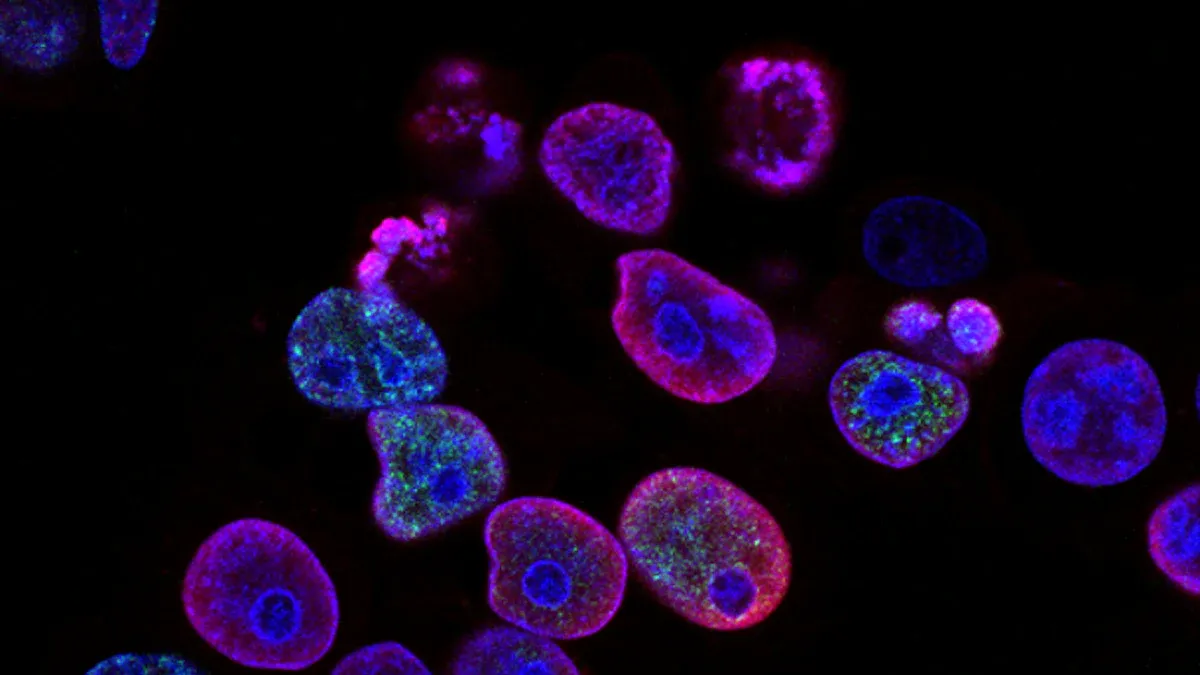
Cell Viability Assessment
You need to check if your cells stay healthy after exposure to the extracts. You can use special tests called assays. These tests help you measure how many cells survive. Two common assays are the MTT assay and the Neutral Red Uptake assay. Each test uses a different method to show cell health.
Here is a table that explains these assays:
Assay Type | Description | Cell Line Used |
|---|---|---|
Neutral Red Uptake | Evaluates cell viability based on the uptake of neutral red dye by living cells. | BALB/c 3T3 |
MTT Assay | Assesses mitochondrial metabolic activity by measuring the reduction of MTT dye to formazan. | BALB/c 3T3 |
You can use these assays to see if your PET heat shrink tubing extracts harm the cells. If the cells take up the dye or change color, they are alive. If they do not, the extract may be toxic.
Tip: Always run your assays with both positive and negative controls. This helps you know if your test works as it should.
Data Interpretation and Acceptance Criteria
After you finish your assays, you need to look at your results. You can compare the number of living cells in your test wells to the number in your control wells. If your test wells have at least 70% cell survival compared to the negative control, your sample passes the cytotoxicity testing for PET heat shrink ISO 10993-5.
You should record your results in a clear table or chart. Write down the percent of cell survival for each sample. If your positive control shows low survival, and your negative control shows high survival, your test is valid.
You can use these steps to decide if your PET heat shrink tubing is safe:
Check cell survival in each well.
Compare test wells to controls.
Look for at least 70% survival in test wells.
Record and report your findings.
Note: If your sample does not meet the acceptance criteria, you may need to repeat the test or check your sample preparation.
Documentation and Reporting
Recording Procedures and Results
You need to keep clear records during cytotoxicity testing for PET heat shrink tubing. Good notes help you show you did each step right. Write down what you do, like sample prep, extraction, cell culture, and assay results. Use a notebook or computer log to write dates, times, and who did each step. If you see any changes or problems, write those down too.
Tip: Always put the right name and date on your samples and containers. This helps you not mix things up and keeps your data neat.
You can use tables to show your test results. For example, you can show if your PET heat shrink tubing passed the cytotoxicity test for ISO 10993-5: 2009.
Test Type | Standard | Result |
|---|---|---|
Cytotoxicity Test | ISO 10993-5: 2009 | Passed |
Keeping records like this helps you prove your testing meets the rules.
Preparing Regulatory Reports
You must write a report for the regulators after you finish testing. Your report should have all the important parts from your study. Start with a short summary of your test methods and materials. Add tables that show your results for cytotoxicity and other safety tests. For example, you can add info about sensitization, hemolysis, thrombosis, and genotoxicity.
Regulatory Test | Result | Impact on Medical Devices |
|---|---|---|
Cytotoxicity | Noncytotoxic | Safe for direct patient contact |
Sensitization | Nonirritant | Reduces risk of allergic reactions |
Hemolysis | Nonhemolytic | Prevents blood cell damage |
Thrombosis | Superior coagulation profiles | Lowers risk of clot formation |
Genotoxicity | No genotoxic effects | Safe for long-term use in medical equipment |
List all steps, controls, and acceptance rules in your report. Add photos or charts if you have them. Make sure your report is easy to read and matches what the regulators want.
Good notes and clear reports help you show your PET heat shrink tubing is safe for medical use. You also make it easier for regulators to check your work and approve your product.
You must use clear steps for cytotoxicity testing of PET heat shrink tubing with ISO 10993-5. Cleanrooms help keep out dust and germs. Using the same methods each time gives you results you can trust. Good notes show your tubing is safe and help you stay out of trouble.
Clean manufacturing helps PET tubing stay safe for people.
Cleanrooms keep samples from getting dirty during tests.
Good records show your tubing is safe for the body.
Regulatory Document | Role |
|---|---|
USP Class VI | Needs tests to check if materials cause bad reactions |
ISO 10993 | Lists rules for biocompatibility |
FDA Title 21 | Checks if device materials are safe and work well |
EU Regulation 1907/2006 | Makes sure chemicals are safe for health |
You should always follow ISO 10993-5 to keep people safe and follow the rules.
FAQ
What is ISO 10993-5 cytotoxicity testing?
You use ISO 10993-5 cytotoxicity testing to check if a material harms living cells. This test helps you see if PET heat shrink tubing is safe for medical use.
Why do you use L-929 mouse fibroblast cells?
You use L-929 cells because they react well to toxic substances. These cells help you see if your PET tubing releases harmful chemicals.
How do you know if your PET heat shrink tubing passes the test?
You look for at least 70% cell survival compared to the negative control. If your tubing meets this level, you can call it noncytotoxic.
What controls do you need for the test?
Control Type | Purpose |
|---|---|
Positive Control | Shows cell death |
Negative Control | Shows cell health |
You need both controls to check if your test works correctly.
See Also
Essential Insights on PET Heat Shrink Tubing for Electronics
Choosing the Right FEP Autoclavable Heat Shrink Tubing
FEP Non-Toxic Heat Shrink Tubing: Key Industrial Insights
Differences Between Medical and Industrial Grade Heat Shrink Tubing
FEP Heat Shrink Tubing Versus Alternatives in Radiation Therapy
